Practice point
Salmonella infections in Canadian children
Posted: Dec 3, 2018 | Updated: Sep 22, 2025
Principal author(s)
Joan L. Robinson; Canadian Paediatric Society, Infectious Diseases and Immunization Committee
Abstract
Non-typhoidal Salmonella (NTS) infections are primarily transmitted by contaminated food or water or contact with carrier animals (particularly reptiles), and present with diarrhea. Antibiotics do not decrease the severity or duration of diarrhea and may increase the incidence of NTS carriage, so they should only be used with suspected or proven bacteremia or invasive infection. Typhoid/paratyphoid fever manifests as bacteremia within 60 days of travel to resource-poor countries and presents with fever and variable abdominal complaints. Therefore, blood cultures are indicated for unexplained fever and a relevant travel history. When blood cultures are positive or when a child is unwell pending blood culture results, ceftriaxone is indicated. A switch to oral antibiotics (usually azithromycin) is often possible after blood cultures have cleared and the child is improved.
Keywords: Non-typhoidal Salmonella; Typhoid/paratyphoid fever
The two distinct types of Salmonella infection are non-typhoidal (NTS) and typhoidal.
The genus Salmonella has two species (enterica and bongori) further divided into seven sub-species that encompass about 2,500 serotypes (also known as serovars or strains), of which only S Typhi and S Paratyphi cause typhoid fever (also known as enteric fever). Of note, S Typhimurium is an NTS despite its name.
Epidemiology and clinical presentation
Non-typhoidal Salmonella infection: A broad range of animals can be asymptomatic NTS carriers [1]. Common sources of human infection are animal contact (especially with reptiles) and consumption of food or water contaminated with NTS of human or animal origin. Common contaminated foods are poultry, eggs (which can be contaminated even when intact, from NTS in the oviduct of the hen), dairy products, ground beef, or produce [1]. Even produce with a thick peel, such as melons, can be a source; contamination presumably occurs when the dirty surface is cut. Sprout seeds can be contaminated, resulting in an entire infected crop [2]. Tomatoes can absorb NTS if soaked in contaminated water [3].
The incubation period is typically 12 h to 48 h but can be up to 7 days [1]. Patients usually present with non-bloody diarrhea, with or without vomiting and fever. Other clinical manifestations are outlined in Table 1. Disseminated disease is most common if the child is immunocompromised, has asplenia or is <3 months old.
Typhoid/paratyphoid fever: Humans are the primary hosts. Infection is usually acquired in Asia (and less commonly in Africa) from water or food contaminated with feces from a carrier or from direct person-to-person spread.
The incubation period is usually 7 to 14 days (range 3 to 60 days) [4]. Typhoid/paratyphoid fever is an invasive infection that manifests as bacteremia and can progress to sepsis and multi-organ failure. The diagnosis is often not considered initially because children typically present with non-specific symptoms of fever and abdominal manifestations. Involvement of the reticuloendothelial system eventually leads to hepatomegaly and/or splenomegaly. A macular rash (rose spots) is noted on the abdomen in about 30% of cases [4]. Other clinical manifestations are outlined in Table 1. Abscess formation at any site is much less common than with NTS infection [5].
| Table 1 - Clinical manifestations of Salmonella infection | ||
| Non-typhoidal | Typhoidal | |
| Asymptomatic infection | Common | Incidence unknown |
| Acute gastroenteritis | Most patients have nausea, vomiting and diarrhea (non-bloody – persists 3 to 7 days) +/− fever. | Most have abdominal pain. Constipation with a history of recent diarrhea is common. |
| Gastrointestinal bleed | Very rare | Occurs in approximately 10% of hospitalized adults and children [4]. |
| Intestinal perforation | Very rare | Rare – most commonly involves the terminal ileum [4]. |
| Chronic carrier (>12 months) | Very rare in children. Gallstones are a risk factor. | Very rare in children. Gallstones are a risk factor. |
| Bacteremia (disseminated disease) | Occurs in about 6% to 8% of cases in all ages [1][2] and rarely progresses to shock. | Detected in about 80% of cases [2] but presumably always present. Progression to shock is more common than with NTS. |
| Central nervous system involvement (disseminated disease) | Meningitis or brain abscess mainly occur in the first year of life. Encephalopathy is rare. | Like NTS, but encephalopathy is more common and brain abscess very rare. |
| Cardiac/vascular involvement (disseminated disease) | Endocarditis or arteritis (primarily in adults). | Myocarditis and endocarditis (rare). |
| Osteomyelitis (disseminated disease) | Mainly with sickle cell anemia. | Rare. |
| Septic arthritis (disseminated disease) | Mainly with sickle cell anemia. | Rare. |
| Reactive arthritis | Rare in children [13]. | Not described. |
| Urinary tract infection | Difficult to distinguish from contaminated urine. Renal abscesses described. | Very rare but can lead to urinary stones [14]. |
| Neonatal infection from perinatal transmission | Rare [15]. | Rare [4]. |
| There are rare reports of anemia, disseminated intravascular coagulopathy and pulmonary, muscle/soft tissue, hepatobiliary, splenic or genital involvement, primarily in adults with typhoidal infection [4]. | ||
Diagnosis
Non-typhoidal Salmonella: Stool cultures are usually positive and should be submitted for all children with bloody diarrhea or persistent or severe non-bloody diarrhea.
Blood cultures should be submitted when stool yields NTS and the child is febrile or immunocompromised. The inflammatory response to Salmonella bacteremia in infants can be minimal [4]. Therefore, blood cultures are also recommended for afebrile children <3 months [6] and possibly up to 6 months of age who have NTS in stool.
Typhoid/paratyphoid fever: Blood cultures should be submitted for all children with onset of unexplained fever within 2 months of returning from a resource-poor country. Bacteremia is typically low-grade, such that collecting an adequate volume of blood and submitting two blood cultures will increase yield. Stool cultures are only positive in about 30% of patients [4] because the gut infection has often resolved by the time they present. Patients with positive stools but negative blood cultures were presumably all bacteremic at some point.
Management
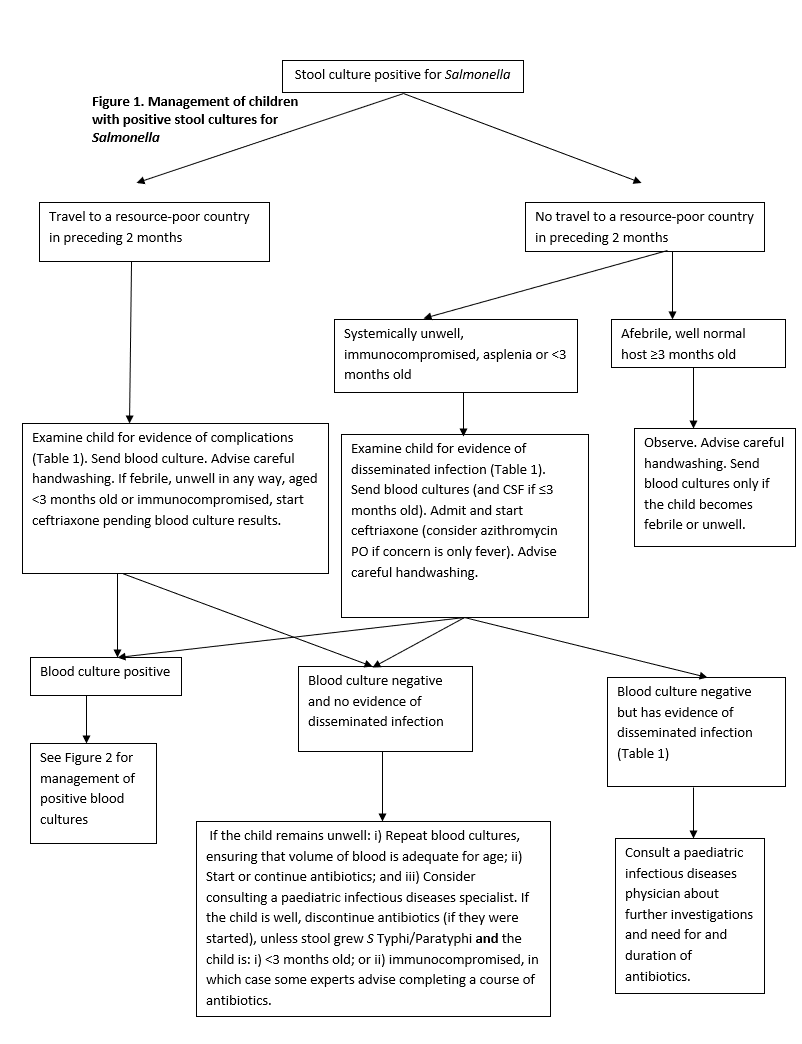
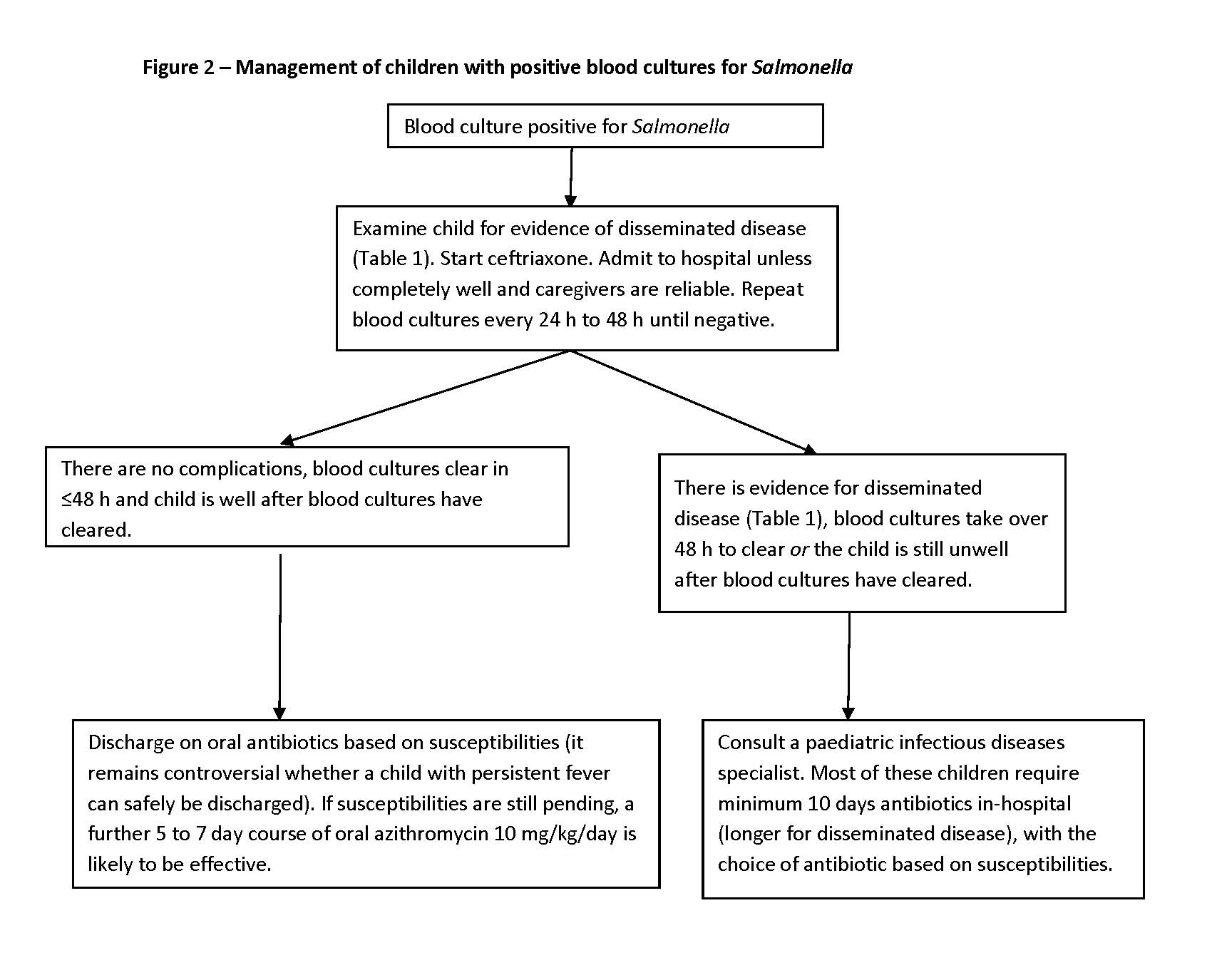
NTS – See Figures 1 and 2 for initial management. Further points are as follows:
- Antibiotics do not decrease the severity or duration of diarrhea and may increase the incidence of carriage, so they should only be used with suspected or proven bacteremia or invasive infection.
- Published studies using azithromycin are limited to use in typhoid/paratyphoid fever, but given its high efficacy and simplicity of administration, azithromycin use for NTS bacteremia is also recommended [7] (Figure 2).
- Patients with diarrhea should be considered infectious. They should not work as food handlers until symptoms fully resolve. Regulations regarding child-care attendance while still symptomatic vary by jurisdiction. There is never a need to document when stools become negative for NTS; asymptomatic carriage often persists for months in young children, but asymptomatic carriers should not be restricted from participating in any activities.
- To find the source and therefore prevent further cases in the same household, the family should be questioned about exposure to animal “treats” that contain raw meat. They should be advised to remove reptiles from the home or at least ensure that the reptile is never in the kitchen or the bathtub.
- Public health is automatically notified of all positive cultures to expedite recognition of outbreaks. The species of Salmonella is a clue to origin.
Typhoid/paratyphoid fever – See Figures 1 and 2 for initial management. Further points are as follows:
- Until recently, ciprofloxacin was commonly used as step-down therapy. Although not approved for pre-pubertal children, ciprofloxacin provides an oral option with intracellular killing and ispostulated to be more effective than ampicillin, cefixime [4] or trimethoprim-sulfamethoxazole. However, resistance is now common and quinolones should be used only when isolates are reported to be susceptible. Azithromycin has become the drug of choice because it, too, provides intracellular killing [8] and there is far less resistance to it.
- The duration of antimicrobials remains controversial. The Canadian Committee to Advise on Tropical Medicine and Travel (CATMAT’s) recommendations for uncomplicated typhoid/paratyphoid fever are 7 days for azithromycin, 3 to 7 days for quinolones, 7 to 14 days for cefixime, 10 to 14 days for ceftriaxone and 14 days for amoxicillin or TMP-SMX [9]. The total duration is unclear for patients who are stepped down to oral therapy.
- Relapse occurs in up to 17% of cases, typically within 4 weeks [10]. Relapse is postulated to be due to residual disease in the reticuloendothelial system rather than antibiotic resistance. Relapse rates may be lower with a 5 to 7 day course of azithromycin than with quinolones or ceftriaxone [11], presumably because azithromycin has excellent tissue and intracellular penetration and a long half-life [4]. Optimal management of relapses is not clear.
- Fever typically persists 6 to 8 days from the start of antibiotics [4]. Fever is not a contraindication to switch to oral antibiotics or to hospital discharge, providing the child is otherwise well.
- Provincial/territorial regulations vary but, in general, food handlers, health care workers and diapered children must have two or three negative stool tests 24 h apart over a variable number of days following completion of antibiotics before returning to work or to child care. Urine cultures may be required if patients have been in an area endemic for schistosomiasis, because Salmonella-Schistosoma binding promotes long-term survival of S Tyhpi or S Paratyphi, with intermittent bacteriuria [12].
Prevention
Non-typhoidal Salmonella: Safe handling of food in the home and good handwashing after contact with animals (especially baby poultry, reptiles and frogs) are key. Some experts recommend that preschool-aged children have no contact with these animals. Immunization of hens in the United Kingdom appears to have had a major impact [10] but remains voluntary in Canada.
Typhoid/paratyphoid fever: Careful food and beverage selection and good hand hygiene during travel to resource-poor countries is vital (http://www.phac-aspc.gc.ca/tmp-pmv/catmat-ccmtmv/diarrhea-diarrhee-eng.php#a52a).
All children ≥24 months old travelling to South Asia (Afghanistan, Bangladesh, Bhutan, India, Nepal, Maldives, Pakistan and Sri Lanka) should receive typhoid vaccine [12]. There is no licensed vaccine for younger children. Immunization should be considered for travellers to other resource-poor countries when they are likely to be exposed to contaminated food or water or when they have achlorhydria, asplenia or sickle cell disease or are immunocompromised. Household contacts of carriers should also be immunized.
The following vaccines are available in Canada (see www.phac-aspc.gc.ca/publicat/cig-gci/p04-typh-eng.php for details on dosing):
- Oral, live vaccine: Vivotif – licensed for children age 5 years and up, with a booster recommended after 7 years.
- Parenteral inactivated vaccine: TYPHERIX or TYPHIM Vi – licensed for age 2 years and up, with a booster recommended after 3 years.
- Parenteral inactivated vaccine combined with hepatitis A vaccine: ViVAXIM – licensed for age 16 years and up, with a typhoid booster recommended after 3 years.
All three vaccines have only about 50% efficacy for S Typhi. Only oral vaccine is thought to provide some protection against S Paratyphi.
|
|
|
|
Addendum
Although most healthy persons with nontyphoidal Salmonella (NTS) gastroenteritis do not require antimicrobial therapy, the AAP’s Red Book (2024 edition)[7] recommends antibiotics for individual who are more prone to developing invasive disease, including those with immature or compromised immune systems. Particularly vulnerable are infants <3 months of age, children with sickle cell disease, HIV, or primary immunodeficiencies, and individuals with a malignancy or an underlying chronic gastrointestinal disease. Based on the increasing prevalence of XDR Salmonella in Pakistan and Iraq, physicians should consider empiric treatment with ceftriaxone or azithromycin for travellers with typhoid fever coming from these countries.
Acknowledgements
This practice point was reviewed by the Acute Care and Community Paediatrics Committees of the Canadian Paediatric Society. It was also reviewed by representatives of the Association of Medical Microbiology and Infectious Disease (AMMI) Canada, with thanks.
CANADIAN PAEDIATRIC SOCIETY INFECTIOUS DISEASES AND IMMUNIZATION COMMITTEE
Members: Michelle Barton-Forbes MD; Natalie A Bridger MD; Shalini Desai MD; Michael Forrester MD; Ruth Grimes MD (Board Representative); Charles Hui (past member); Nicole Le Saux MD (Chair); Joan L Robinson MD (past Chair); Otto G Vanderkooi MD (past member)
Liaisons: Upton D Allen MBBS, Canadian Paediatric & HIV/AIDS Research Group; Tobey Audcent MD, Committee to Advise on Tropical Medicine and Travel (CATMAT), Public Health Agency of Canada; Carrie Byington MD, Committee on Infectious Diseases, American Academy of Pediatrics; Fahamia Koudra MD, College of Family Physicians of Canada; Howard Njoo MD, Public Health Agency of Canada; Marc Lebel MD, IMPACT (Immunization Monitoring Program, ACTIVE); Jane McDonald MD, Association of Medical Microbiology and Infectious Disease Canada; Dorothy L Moore MD, National Advisory Committee on Immunization (NACI)
Consultant: Noni E. MacDonald MD
Principal author: Joan L. Robinson MD
Updated (September 2025): Michelle Barton MD, Sergio Fanella MD, Isabelle VielTheriault MD
References
- Pegues DA, Miller SI. Salmonella species. In: Bennett JE, Dolin R, Blaser MJ. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases, 8th edn. Philadelphia, PA: Elsevier, 2015:2559-68.
- Sadler-Reeves L, Aird H, de Pinna E, et al;. The occurrence of Salmonella in raw and ready-to-eat bean sprouts and sprouted seeds on retail sale in England and Northern Ireland. Lett Appl Microbiol 2016;62(2):126-9.
- van Dyk BN, de Bruin W, du Plessis EM, Korsten L. Microbiological food safety status of commercially produced tomatoes from production to marketing. J Food Prot 2016;79(3):392-406.
- Crump JA, Sjölund-Karlsson M, Gordon MA, Parry CM. Epidemiology, clinical presentation, laboratory diagnosis, antimicrobial resistance, and antimicrobial management of invasive Salmonella infections. Clin Microbiol Rev 2015;28(4):901-37.
- Harris JB, Ryan ED. Enteric fever and other causes of fever and abdominal symptoms. In: Bennett JE, Dolin R, Blaser MJ. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases, 8th edn. Philadelphia, PA: Elsevier, 2015:1270-82.
- Geme JW, Hodes HL, Marcy SM, et al. Consensus: Management of Salmonella infection in the first year of life. Pediatr Infect Dis J 1988;7(9):615-21.
- American Academy of Pediatrics. [Salmonella infections] In: Kimberlin DW, Banerjee R, Barnett ED, Lynfield R, Sawyer MH, eds. Red Book: 2024 Report of the Committee on Infectious Diseases. Itaska, IL: American Academy of Pediatrics; 2024:742-9
- Trivedi NA, Shah PC. A meta-analysis comparing the safety and efficacy of azithromycin over the alternate drugs used for treatment of uncomplicated enteric fever. J Postgrad Med 2012;58(2):112-8.
- Public Health Agency of Canada. Statement on International Travellers and Typhoid: An Advisory Committee Statement (ACS) Committee to Advise on Tropical Medicine and Travel (CATMAT). February 2014 (Accessed Sept 29, 2025).
- Bula-Rudas FJ, Rathore MH, Maraqa NF. Salmonella infections in childhood. Adv Pediatr 2015;62(1):29-58.
- Butler T. Treatment of typhoid fever in the 21st century: Promises and shortcomings. Clin Microbiol Infect 2011;17(7):959-63.
- Barnhill AE, Novozhilova E, Day TA, Carlson SA. Schistosoma-associated Salmonella resist antibiotics via specific fimbrial attachments to the flatworm. Parasit Vectors 2011;28;4:123.
- Keithlin J, Sargeant JM, Thomas MK, Fazil A. Systematic review and meta-analysis of the proportion of non-typhoidal Salmonella cases that develop chronic sequelae. Epidemiol Infect 2015;143(7):1333-51.
- Değirmenci T, Arı A, Kozacıoğlu Z, Örs B, Günlüsoy B. Salmonella typyhi isolated from urine culture before percutaneous nephrolithotomy: A case report. Urol J 2014;11(1):1350-2.
- Rai B, Utekar T, Ray R. Preterm delivery and neonatal meningitis due to transplacental acquisition of non-typhoidal Salmonella serovar montevideo. BMJ Case Rep 2014;2014:pii: bcr2014205082.
Disclaimer: The recommendations in this position statement do not indicate an exclusive course of treatment or procedure to be followed. Variations, taking into account individual circumstances, may be appropriate. Internet addresses are current at time of publication.